Regulatory-grade AI for submission-ready documents
Yseop generates regulatory documents you can defend under audit. Built for accuracy, traceability, and repeatability in GxP-regulated environments.
Yseop generates regulatory documents you can defend under audit. Built for accuracy, traceability, and repeatability in GxP-regulated environments.

Content generation is easy. Getting content approved is not. Most AI tools stop at drafting. Regulatory teams need content that is grounded, traceable, and consistent across submissions.

Create submission-ready drafts from structured and narrative data. Ground every draft in source data from the start.
Coordinate drafting, validation, and review across teams. Update once. Apply everywhere—across documents and submissions
Work directly in Microsoft Word and connect with Veeva Vault. Built-in traceability, audit trails, and QC checks for inspection readiness.
See why teams choose Yseop over other AI tools

|
Competitors Others | |
|---|---|---|
|
Accurate — not just generated
Grounded in your source data and validated against regulatory rules and templates
|
||
|
Traceable — every claim accounted for Every sentence is linked to its source from the start Full audit trail — ready for inspection at any time |
||
|
Built for GxP environments Designed and validated for regulatory requirements—not adapted after the fact |
||
|
Proven in production Deployed across Top 10 global pharma companies. Used in 300+ clinical trials. Submissions approved by FDA and EMA. |
||
|
Human control at every step AI executes. Your team reviews, validates, and approves |
Yseop is trusted by the world’s leading life sciences organizations because it generates content they can stand behind—and submit with confidence.

Our AI-powered NLG platform speeds up drug market timelines and eliminates human error in data review.
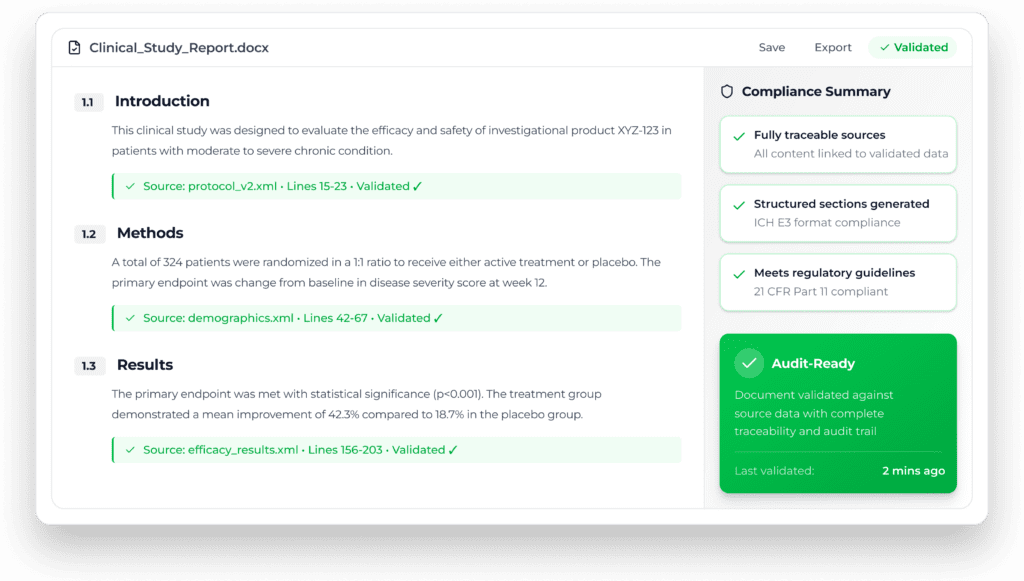
Yseop transforms structured datasets into fully formatted regulatory documents—ensuring consistency, traceability, and compliance across every output.
Yseop transforms structured datasets into fully formatted regulatory documents—ensuring consistency, traceability, and compliance across every output.
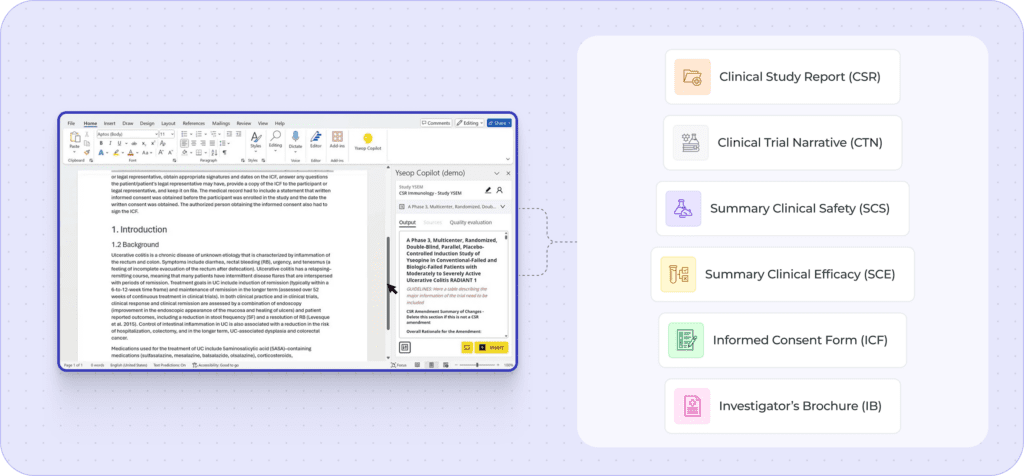
Connect to SDTM, ADaM, and clinical data repositories without manual data extraction.
AI understands regulatory structure and clinical context, not just text patterns.
Every sentence validated against source data with complete audit documentation.




What to know before using AI in regulatory submissions
Regulatory-grade AI is designed for GxP-regulated environments, where every document must be accurate, traceable, and consistent.
Unlike general-purpose AI, it includes built-in validation, audit trails, and controls to support regulatory review and submission.
Most AI tools generate text. Yseop generates content that is:
It is built specifically for regulatory workflows—not adapted from generic AI.
Yes—if it meets regulatory standards.
Yseop is used in production by top global pharma companies and has supported submissions approved by FDA and EMA.
Every output is validated, traceable, and reviewed by your team before submission.
Yseop combines data-driven generation with built-in validation:
Let’s start automating your data to narratives today!








© 2026 YSEOP. All rights reserved.